Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Missyou Chenmical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shmychem.com | |||
 | +86 (21) 5858-3907 | |||
 | +86 (21) 5858-3907 | |||
 | doreen@shmychem.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Chem Service, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemservice.com | |||
 | +1 (610) 692-3026 | |||
 | +1 (610) 692-8729 | |||
 | info@chemservice.com | |||
| Chemical manufacturer since 1962 | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Jiangyin Trust-Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.trust-chem.com | |||
 | +86 (510) 8625-7670 8626-8020 8626-9706 | |||
 | +86 (510) 8626-8021 | |||
 | trust@hi2000.com | |||
| Chemical manufacturer since 1996 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Alcohols, phenols, phenolic compounds and derivatives >> Phenol and its halogenated, sulfonated, nitrated or nitrosated derivatives |
|---|---|
| Name | Thiophenol |
| Synonyms | Benzenethiol; Phenyl mercaptan |
| Molecular Structure | 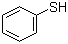 |
| Molecular Formula | C6H6S |
| Molecular Weight | 110.18 |
| CAS Registry Number | 108-98-5 |
| EC Number | 203-635-3 |
| FEMA | 3616 |
| SMILES | C1=CC=C(C=C1)S |
| Density | 1.078 |
|---|---|
| Melting point | -15 °C |
| Boiling point | 167-169 °C |
| Refractive index | 1.588-1.59 |
| Flash point | 50 °C |
| Water solubility | insoluble |
| Hazard Symbols |      GHS02;GHS05;GHS06;GHS07;GHS09 Danger Details GHS02;GHS05;GHS06;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H300-H301-H310-H311-H314-H315-H318-H330-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P262-P264-P264+P265-P270-P271-P273-P280-P284-P301+P316-P301+P330+P331-P302+P352-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P320-P321-P330-P332+P317-P361+P364-P362+P364-P363-P370+P378-P391-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2337 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Thiophenol, also known as mercaptobenzene or benzenethiol, is a chemical compound with the chemical formula C₆H₅SH. It is characterized by a benzene ring bonded to a sulfur atom and a thiol (-SH) functional group. Thiophenol was first synthesized in the 19th century, when chemists were exploring the properties and reactions of thiols and aromatic compounds. Its discovery marked an important development in organic chemistry, especially in the understanding of sulfur-containing organic molecules. Thiophenol consists of a benzene ring (C₆H₅-) to which is attached a sulfur atom (-S-) and a hydrogen atom (-H). It is a colorless to pale yellow liquid with a strong malodor reminiscent of rotting cabbage, which is typical of thiol compounds. Thiophenol is widely used as a building block in organic synthesis, especially in the production of pharmaceuticals, agrochemicals, and dyes. Its sulfur-containing functional group promotes a variety of chemical reactions, including nucleophilic substitution and metal complex formation. It is a key intermediate in the synthesis of other sulfur-containing compounds such as thiophenolates and disulfides. These intermediates can be used to make rubber additives, pesticides, and antioxidants. The reactivity and stability of thiophenols make them valuable in industrial processes, including the production of specialty chemicals and polymer additives. It helps modify polymer surfaces and synthesize materials with specific chemical and physical properties. Thiophenols are toxic if inhaled, ingested, or absorbed through the skin. Proper handling and ventilation are essential to prevent exposure to its vapor or liquid form. The strong odor of thiophenols is easily detectable even at low concentrations and serves as a warning of potential exposure. Thiophenols and their derivatives are biodegradable in soil and water and have reduced environmental persistence compared to some other aromatic compounds. Due to their toxicity and environmental impact, the use and disposal of thiophenols are regulated in different jurisdictions to minimize potential hazards. References 2018. XPS Analysis of 2- and 3-Aminothiophenol Grafted on Silicon (111) Hydride Surfaces. Molecules. DOI: 10.3390/molecules23102712 2013. Peptide Release, Side-Chain Deprotection, Work-Up, and Isolation. Methods in Molecular Biology. DOI: 10.1007/978-1-62703-544-6_3 2003. Mechanistic Studies of the Tellurium(II)/Tellurium(IV) Redox Cycle in Thiol Peroxidase-like Reactions of Diorganotellurides in Methanol. Journal of the American Chemical Society. DOI: 10.1021/ja029590m |
| Market Analysis Reports |