Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Shanghai Sinofluoro chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinofluoro.com | |||
 | +86 (21) 6427-9360 | |||
 | +86 (21) 6427-8603 | |||
 | simon.lv@sinofluoro.com jerry.chen@sinofluoro.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hangzhou Rongda Pharm & Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rdpharm.com | |||
 | +86 (571) 8808-8387 8808-6013 | |||
 | +86 (571) 8808-8387ex 802 | |||
 | sales@rdpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Lianyungang United Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lyg-china.com | |||
 | +86 (518) 8569-2709 | |||
 | +86 (518) 8501-5182 | |||
 | service@lyg-china.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Nanjing Yingkun Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yingkunchem.com | |||
 | +86 (25) 8341-1660 | |||
 | +86 (25) 8321-2803 | |||
 | isabela@yingkunchem.com hefengrili@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Zhejiang Warrant Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhejiang-warrant.com | |||
 | +86 (512) 8518-0611 +86 17312581805 | |||
 | +86 (512) 8917-1181 | |||
 | sales@zhejiang-warrant.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shaoxing Fangxiao Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fangxiaochem.com | |||
 | +86 (571) 2829-7060 +86 18969095308 | |||
 | +86 (571) 2829-7065 | |||
 | sales@fangxiaochem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Central Glass Trading (Shanghai) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cgco.com.cn | |||
 | +86 (21) 6219-9791 | |||
 | +86 (21) 6215-2691 | |||
 | sales@cgco.com.cn | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shanghai Weiyuan Technology Co., Ltd | China | |||
|---|---|---|---|---|
 | www.shweiyuan.cn | |||
 | +86 13024122785 | |||
 | sales@shweiyuan.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2021 | ||||
| P and M Invest Ltd. | Russia | |||
|---|---|---|---|---|
 | www.fluorine.ru | |||
 | +7 (495) 135-6494 | |||
 | +7 (495) 135-6509 | |||
 | sales@fluorine1.ru | |||
| Chemical manufacturer | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty alcohol |
|---|---|
| Name | 4,4,5,5,5-Pentafluoro-1-pentanol |
| Synonyms | 4,4,5,5,5-Pentafluoropentanol |
| Molecular Structure | 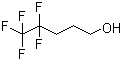 |
| Molecular Formula | C5H7F5O |
| Molecular Weight | 178.10 |
| CAS Registry Number | 148043-73-6 |
| EC Number | 421-360-9 |
| SMILES | C(CC(C(F)(F)F)(F)F)CO |
| Density | 1.35 g/mL (Expl.) |
|---|---|
| Boiling point | 62-64 °C (35 mmHg) (Expl.) |
| Refractive index | 1.334 (Expl.) |
| Flash point | 63 °C (Expl.) |
| Hazard Symbols |   GHS02;GHS07 Warning Details GHS02;GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H302+H412 Details | ||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P264-P270-P273-P280-P301+P317-P303+P361+P353-P330-P370+P378-P403+P235-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
4,4,5,5,5-Pentafluoro-1-pentanol is a fluorinated alcohol that has gained attention in both industrial and research applications due to its unique chemical properties. As a highly fluorinated compound, it possesses characteristics that differentiate it from other alcohols, particularly its enhanced stability and solvent properties. The compound is part of a broader group of fluorinated alcohols, which are widely used in the chemical industry for various purposes, such as solvents, surfactants, and intermediates in the synthesis of more complex compounds. The discovery of 4,4,5,5,5-pentafluoro-1-pentanol can be traced back to ongoing research into fluorinated compounds, which have become important in a variety of sectors due to their distinctive physical and chemical properties. Fluorine, being highly electronegative, imparts unique characteristics to molecules, such as increased resistance to chemical degradation, enhanced thermal stability, and solubility in non-polar solvents. These properties make fluorinated alcohols, including 4,4,5,5,5-pentafluoro-1-pentanol, valuable in many specialized applications, particularly in areas where stability and chemical inertness are required. In terms of its structure, 4,4,5,5,5-pentafluoro-1-pentanol consists of a pentanol backbone with five fluorine atoms attached to the 4 and 5 positions of the carbon chain. This arrangement significantly alters the compound's chemical behavior compared to non-fluorinated alcohols. The presence of fluorine atoms contributes to its low reactivity with other chemicals, making it useful in applications where reactions with other substances need to be minimized. One of the primary applications of 4,4,5,5,5-pentafluoro-1-pentanol is in the field of materials science, particularly in the development of advanced coatings and surface treatments. Its ability to form stable films on surfaces makes it a useful component in coatings that require resistance to chemicals, high temperatures, and moisture. Additionally, its unique solubility properties allow it to function as a solvent in various chemical reactions, especially in systems that involve polar and non-polar components. In the pharmaceutical industry, 4,4,5,5,5-pentafluoro-1-pentanol has potential uses as a solvent in the formulation of drugs or as an intermediate in the synthesis of fluorinated pharmaceuticals. Fluorinated compounds have become increasingly important in drug design, as the incorporation of fluorine atoms can improve the biological activity, stability, and bioavailability of pharmaceutical agents. The ability of 4,4,5,5,5-pentafluoro-1-pentanol to interact with other fluorinated compounds opens up possibilities for creating new classes of drugs with enhanced properties. Another significant application of this compound is in the development of advanced lubricants and oils. Fluorinated alcohols, such as 4,4,5,5,5-pentafluoro-1-pentanol, are known for their excellent thermal stability and resistance to oxidative degradation, which make them ideal for use in harsh environments where conventional lubricants would fail. This includes high-performance applications in automotive, aerospace, and industrial machinery. Although 4,4,5,5,5-pentafluoro-1-pentanol is a relatively specialized compound, its broad range of applications in various industries underscores its importance in advancing technologies that require highly stable and versatile chemicals. The compound’s properties make it particularly valuable in the development of next-generation materials, pharmaceuticals, and industrial products. In conclusion, 4,4,5,5,5-pentafluoro-1-pentanol represents a class of fluorinated alcohols that provide unique advantages in stability, solubility, and resistance to chemical degradation. Its applications span multiple industries, from pharmaceuticals to materials science, where it serves as an important component in the development of advanced products and technologies. References 2020. Systematic Investigation of Lipophilicity Modulation by Aliphatic Fluorination Motifs. Journal of Medicinal Chemistry, 63, 1. DOI: 10.1021/acs.jmedchem.9b01172 2011. Incorporation of Fluorous Glycosides to Cell Membrane and Saccharide Chain Elongation by Cellular Enzymes. Topics in current chemistry, 308, 1. DOI: 10.1007/128_2011_276 |
| Market Analysis Reports |