Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Minakem S.A.S. | France | |||
|---|---|---|---|---|
 | www.minakem.com | |||
 | +33 (3) 2064-6830 | |||
 | +33 (3) 2064-3198 | |||
 | contact@minakem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shen Zhen Reagent Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.haoreagent.com | |||
 | +86 (755) 8945-9231 | |||
 | sales@haoreagent.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Tongxiang Hengda Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hengdachem.com | |||
 | +86 (573) 8828-3621 8871-1709 | |||
 | +86 (573) 8822-3196 / 8872-1305 | |||
 | sales@hengdachem.com | |||
| Chemical manufacturer since 2009 | ||||
| Classification | Organic raw materials >> Inorganic acid ester |
|---|---|
| Name | Diethyl pyrocarbonate |
| Synonyms | DEPC; Diethyl oxydiformate; Ethoxyformic anhydride; Pyrocarbonic acid diethyl ester |
| Molecular Structure | 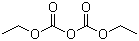 |
| Molecular Formula | C6H10O5 |
| Molecular Weight | 162.14 |
| CAS Registry Number | 1609-47-8 |
| EC Number | 216-542-8 |
| SMILES | CCOC(=O)OC(=O)OCC |
| Density | 1.101 |
|---|---|
| Boiling point | 93-94 °C (18 mmHg) |
| Refractive index | 1.396-1.4 |
| Flash point | 69 °C |
| Water solubility | slow decomposition |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
Diethyl pyrocarbonate (DEPC) is an organic chemical compound first synthesized in the mid-20th century and has since become a valuable reagent in biochemical research and industrial applications. It is a diester of pyrocarbonic acid and is commonly used as a potent agent for modifying biological molecules. One of the primary applications of DEPC is in molecular biology, where it is utilized to modify nucleic acids and proteins. DEPC acts as a potent inactivator of ribonucleases (RNases), enzymes that degrade RNA. By reacting with the histidine residues of RNases, DEPC effectively deactivates these enzymes, which is crucial for the preservation of RNA samples in research laboratories. This application is particularly important during RNA extraction and purification processes, where the presence of active RNases can lead to the degradation of RNA, thereby compromising experimental results. In addition to its role in molecular biology, DEPC is used in the chemical industry for various purposes. It serves as a reagent in the synthesis of other chemical compounds, particularly those involving esterification reactions. Its reactivity with alcohols and phenols makes it useful for creating esters and other derivatives, which are employed in the production of pharmaceuticals, agrochemicals, and specialty chemicals. DEPC is also used as a cross-linking agent in the preparation of certain polymer materials. Its ability to form stable ester bonds allows for the modification of polymer structures, enhancing their properties and performance in specific applications. This feature makes DEPC valuable in the development of advanced materials with tailored characteristics. Despite its usefulness, DEPC must be handled with care due to its potential health hazards. It is a toxic and irritant substance, requiring appropriate safety measures and protective equipment during its use. Research continues to focus on improving the safety protocols associated with DEPC and exploring alternative reagents with similar functionalities but reduced toxicity. References 1979. Chemical modification of histidyl and lysyl residues in yeast enolase. Biochimica et Biophysica Acta (BBA) - Enzymology, 578(1), 1-9. DOI: 10.1016/0005-2744(79)90081-0 1997. Basic Nucleic Acid Procedures. Molecular Diagnostics, 1, 29-45. DOI: 10.1007/978-1-4757-2588-9_3 1991. Modification of system A amino acid carrier by diethyl pyrocarbonate. The Journal of Biological Chemistry, 266(2), 987-992. DOI: 10.1016/s0021-9258(17)35243-2 |
| Market Analysis Reports |