Online Database of Chemicals from Around the World
| Shandong Hongkun Group | China | |||
|---|---|---|---|---|
 | www.hongkungroup.com | |||
 | +86 (718) 395-3311 | |||
 | +86 (718) 461-7902 | |||
 | mwu59@yahoo.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Jiangsu Suoteng New Material Technology Co, Ltd. | China | |||
|---|---|---|---|---|
 | www.jssunton.com | |||
 | +86 18915258369 | |||
 | sales@suntonchem.com | |||
| Chemical distributor since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Metropolitan Eximchem Ltd. | India | |||
|---|---|---|---|---|
 | www.metrosun.com | |||
 | +91 (22) 2408-1528 | |||
 | +91 (22) 2407-7375 | |||
 | info@metrosun.com | |||
| Chemical manufacturer since 1996 | ||||
| Wuxi Zhufeng Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.luquchem.com | |||
 | +86 (510) 8395-2589 +86 13606190291 | |||
 | +86 (510) 8395-7198 | |||
 | sales@luquchem.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antibiotics >> Other antibiotics |
|---|---|
| Name | 2,3-Dimethyl-2,3-diphenylbutane |
| Synonyms | 1,1'-(1,1,2,2-Tetramethylethylene)dibenzene; 1,1'-(1,1,2,2-Tetramethyl-1,2-ethanediyl)bisbenzene |
| Molecular Structure | 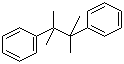 |
| Molecular Formula | C18H22 |
| Molecular Weight | 238.37 |
| CAS Registry Number | 1889-67-4 |
| EC Number | 217-568-2 |
| SMILES | CC(C)(C1=CC=CC=C1)C(C)(C)C2=CC=CC=C2 |
| Melting point | 90-110 °C |
|---|---|
| Water solubility | insoluble |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H317-H413 Details | ||||||||||||||||||||
| Safety Statements | P261-P272-P273-P280-P302+P352-P321-P333+P317-P362+P364-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
2,3-Dimethyl-2,3-diphenylbutane (DMDPB) is an organic compound used as a free radical initiator in chemical synthesis. It has two tert-butyl groups attached to a central carbon, each of which is bonded to a phenyl group, making it highly stable and efficient in generating free radicals. DMDBB was first synthesized in the mid-20th century as part of the quest for stable free radical initiators. Researchers looked for compounds that could decompose to generate free radicals under controlled conditions without significant degradation. The synthesis of DMDPB involves the coupling of 2,3-dimethylbutane to a benzene ring, resulting in a stable crystalline compound. Its unique structure and stability make it ideal for free radical chemistry applications. The primary use of DMDPB is as a free radical initiator for polymerization reactions. It is particularly effective in initiating the polymerization of monomers such as styrene, methyl methacrylate, and acrylates. DMDPB decomposes under heating to generate free radicals that help form polymer chains, resulting in high-quality polymers with controlled molecular weight and properties. In synthetic organic chemistry, DMDPB is used to generate free radicals for a variety of reactions, including hydrocarbon functionalizations and free radical coupling processes. Its controlled decomposition is valuable in reactions that require precise free radical generation. DMDPB is used to study free radical mechanisms and kinetics. Its stability allows for detailed examination of free radical behavior under different conditions, providing insight into free radical chemistry and aiding in the development of new synthetic methods. Due to its high thermal stability, DMDPB is sometimes used as a stabilizer in high temperature chemical processes. Its presence helps maintain the desired chemical environment by scavenging unwanted free radicals and preventing side reactions. DMDPB is generally stable but should be handled with care, especially at elevated temperatures or in the presence of strong oxidants. Protective measures such as gloves and goggles are recommended when handling the compound to prevent contact with skin or eyes. References 2016. Nanostructured Ionomeric Elastomers. Designing of Elastomer Nanocomposites: From Theory to Applications, . DOI: 10.1007/12_2016_8 2023. Highly reactive polyisobutylene through cationic polymerization of isobutylene. Journal of Polymer Research, . DOI: 10.1007/s10965-023-03706-6 |
| Market Analysis Reports |