Online Database of Chemicals from Around the World
| Shanghai Nanxiang Reagent Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nx-reagent.com | |||
 | +86 (21) 3912-7448 +86 13512139665 | |||
 | +86 (21) 3912-7431 | |||
 | xumiao@nx-reagent.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| SRC Laboratories Pvt Ltd | India | |||
|---|---|---|---|---|
 | www.srclaboratories.com | |||
 | +91 (40) 2307-7999 | |||
 | +91 (40) 2307-7999 | |||
 | info@srclaboratories.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2011 | ||||
| LOBA Feinchemie AG | Austria | |||
|---|---|---|---|---|
 | www.loba.co.at | |||
 | +43 (223) 277-391 | |||
 | +43 (223) 276-677 | |||
 | sales@loba.co.at | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Cloudscape Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cloudscapechem.com | |||
 | +86 (21) 6630-2580 | |||
 | +86 (21) 6631-4011 | |||
 | mollyqin@cschemsh.com molly0919@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Suqian Nanxiang Chemicals Manufacturing Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nanxiangchem.com | |||
 | +86 (0527) 8483-6748 | |||
 | sales@nanxiangchem.com | |||
 | QQ Chat | |||
 | WeChat: 13512139665 | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Organo Fine Chemicals | India | |||
|---|---|---|---|---|
 | www.organo.in | |||
 | +91 (22) 2201-6968 2206-7521 | |||
 | +91 (22) 2205-9299 / 2208-3184 | |||
 | info@organo.in | |||
| Chemical manufacturer | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Jintan Taisheng Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.taishengchem.com | |||
 | +86 (519) 8289-5888 8289-9888 | |||
 | +86 (519) 8289-1888 | |||
 | wqa@yidongchem.com | |||
| Chemical manufacturer | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Amino compound >> Acyclic monoamines, polyamines and their derivatives and salts |
|---|---|
| Name | 2-Bromoethylamine hydrobromide |
| Molecular Structure | 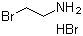 |
| Molecular Formula | C2H6BrN.HBr |
| Molecular Weight | 204.89 |
| CAS Registry Number | 2576-47-8 |
| EC Number | 219-924-2 |
| SMILES | C(CBr)N.Br |
| Melting point | 172-176 °C |
|---|---|
| Water solubility | > 500 g/L (20 °C) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H317-H319-H335-H412 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P272-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
2-Bromoethylamine hydrobromide is an organic compound with the molecular formula C2H6Br2N. It is primarily utilized as a building block in organic synthesis due to its reactivity and versatility. The discovery of 2-bromoethylamine hydrobromide can be traced back to efforts in developing halogenated amines for various applications in organic chemistry. Researchers were interested in halogenated amines due to their potential in nucleophilic substitutions, which are crucial for synthesizing a wide range of compounds. This compound is synthesized through the bromination of ethylamine, followed by the formation of the hydrobromide salt. The reaction involves treating ethylamine with hydrobromic acid and bromine or a brominating agent, resulting in the desired product. The introduction of bromine in the ethylamine structure enhances its reactivity, making it a valuable intermediate in various chemical reactions. 2-Bromoethylamine hydrobromide finds applications in pharmaceutical chemistry, particularly in the synthesis of bioactive compounds. It serves as a versatile building block in the production of various pharmaceuticals, including antihistamines and anti-inflammatory agents. The presence of the bromine atom allows for further functionalization, enabling the synthesis of more complex molecular architectures. In addition to its pharmaceutical applications, 2-bromoethylamine hydrobromide is also utilized in the production of agrochemicals. It acts as a key intermediate in the synthesis of herbicides and pesticides, contributing to the development of effective agricultural products. Its reactivity facilitates the introduction of various functional groups, enhancing the efficacy of these agrochemicals. Furthermore, this compound is employed in the synthesis of polymeric materials. Its ability to act as a crosslinking agent in polymer chemistry allows for the creation of specialized materials with tailored properties. The incorporation of 2-bromoethylamine hydrobromide into polymer formulations can enhance mechanical strength and thermal stability, making it valuable in the development of advanced materials for various industrial applications. The discovery and continued study of 2-bromoethylamine hydrobromide reflect its importance in organic synthesis, pharmaceuticals, and materials science. Its versatility as a reactive intermediate enables researchers to explore new pathways in the synthesis of valuable compounds, contributing to advancements in multiple fields of chemistry. References 2024. Mixed Ligand Metal (II) Complexes from a Functionalized Ionic Liquid Tagged Schiff Base and N-Methyl Imidazole: Synthesis, Spectroscopic Studies and Antibacterial Effects On Rhizosphere Bacteria. Chemistry Africa. DOI: 10.1007/s42250-024-01145-4 2019. A novel cross-linkable, polyaspartamide derivative-containing cinnamoyl groups with temperature and pH dual stimuli-responsiveness. Iranian Polymer Journal, 28(1). DOI: 10.1007/s13726-018-00686-z 1981. S-Aminoethylation of Human Globin Chains Using 2-Bromoethylamine Hydrobromide. Hemoglobin, 5(1). DOI: 10.3109/03630268108991814 |
| Market Analysis Reports |