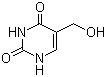
5-Hydroxymethyluracil is a pyrimidine nucleobase derivative in which a hydroxymethyl group is substituted at the 5-position of uracil. Its molecular formula is C5H6N2O3, with a molecular weight of approximately 142.11 g/mol. The uracil core is a planar, electron-deficient heteroaromatic system with keto groups at positions 2 and 4, and the hydroxymethyl group at position 5 introduces both hydrogen-bonding capability and a site for further chemical modification.
The compound can be formed naturally through oxidative modification of thymine residues in DNA or via enzymatic methylation and subsequent hydroxylation pathways. Chemically, it is often synthesized from uracil through selective hydroxymethylation at the 5-position using formaldehyde or other one-carbon donors under controlled conditions. Reaction optimization ensures regioselective functionalization while preserving the keto and ring integrity.
Chemically, 5-hydroxymethyluracil exhibits reactivity at the hydroxymethyl group and the nucleophilic positions of the pyrimidine ring. The hydroxymethyl group can undergo oxidation to form 5-formyluracil or 5-carboxyuracil, esterification, or conjugation with other molecules. The uracil ring participates in hydrogen bonding, tautomerization, and nucleophilic addition reactions, making it reactive in both biochemical and synthetic contexts. Its polar nature and hydrogen-bonding ability significantly influence solubility, intermolecular interactions, and participation in nucleic acid structures.
The compound is generally a crystalline solid at room temperature and is soluble in polar solvents such as water, methanol, and dimethyl sulfoxide. It is chemically stable under neutral conditions but sensitive to strong acids, bases, or oxidizing agents that could modify the hydroxymethyl group or cleave the pyrimidine ring. The combination of the hydroxymethyl functionality and the uracil core allows versatile chemical transformations while maintaining biological relevance.
In practical applications, 5-hydroxymethyluracil serves as a biochemical marker and an intermediate in nucleic acid research. It is studied in the context of DNA epigenetics as an oxidative derivative of 5-methylcytosine, playing a role in DNA demethylation pathways and gene regulation. It is also used in the synthesis of nucleoside analogues, DNA probes, and other modified nucleic acids for research in genetics, enzymology, and therapeutic development.
Overall, 5-hydroxymethyluracil is a functionally versatile pyrimidine derivative with a reactive hydroxymethyl group and a stable heteroaromatic core. Its structural and chemical properties enable selective modification, hydrogen bonding interactions, and applications in nucleic acid chemistry, epigenetic studies, and the design of functional nucleoside analogues.
References
2023. Quantum Computational, Spectroscopic (FT-IR, FT-Raman, NMR, and UV–Vis) Hirshfeld Surface and Molecular Docking-Dynamics Studies on 5-Hydroxymethyluracil (Monomer and Trimer). Molecules, 28(5).
DOI: 10.3390/molecules28052116
2024. Genome-wide distribution of 5-hydroxymethyluracil and chromatin accessibility in the Breviolum minutum genome. Genome Biology.
DOI: 10.1186/s13059-024-03261-3
|































 GHS07 Warning Details
GHS07 Warning Details