Online Database of Chemicals from Around the World
| Jingjiang Sanjing Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sanjingchem.com | |||
 | +86 (519) 8664-4596 | |||
 | +86 (519) 8205-9062 | |||
 | sales@sanjingchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shijiazhuang Haotian Chemcal Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sjzhtchem.com | |||
 | +86 (311) 8568-5134 +86 13383290929 +86 13333118931 | |||
 | +86 (311) 8568-5134 | |||
 | haotianchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhejiang Free Trade Zone Violet Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjviolettech.com | |||
 | +86 (580) 811-8992 | |||
 | +86 (580) 811-9487 | |||
 | jenny@zjviolettech.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Rieke Metals, Inc. | USA | |||
|---|---|---|---|---|
 | www.riekemetals.com | |||
 | +1 (402) 434-2775 | |||
 | +1 (402) 434-2777 | |||
 | sales@riekemetals.com | |||
| Chemical manufacturer | ||||
| Frinton Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.frinton.com | |||
 | +1 (856) 722-7037 | |||
 | +1 (856) 439-1977 | |||
 | sales@frinton.com frinton@frinton.com | |||
| Chemical manufacturer since 1963 | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Jiangxi Liyuan Chemical Tech Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.liyuanchem.com | |||
 | +86 (791) 832-8094 | |||
 | +86 (791) 832-8094 | |||
 | sjpjq001@sina.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Aromatic hydrocarbon |
|---|---|
| Name | 3-Phenyltoluene |
| Synonyms | 3-Methylbiphenyl |
| Molecular Structure | 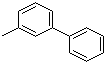 |
| Molecular Formula | C13H12 |
| Molecular Weight | 168.24 |
| CAS Registry Number | 643-93-6 |
| EC Number | 211-404-3 |
| SMILES | CC1=CC(=CC=C1)C2=CC=CC=C2 |
| Density | 1.018 |
|---|---|
| Melting point | 4-5 °C |
| Boiling point | 272 °C |
| Refractive index | 1.601-1.605 |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
3-Phenyltoluene, also known as 1-methyl-3-phenylbenzene, is an aromatic hydrocarbon that has attracted attention due to its structural simplicity and diverse applications in organic synthesis and material science. This compound, featuring a toluene core with a phenyl group attached at the meta position, has been studied for its physicochemical properties and its potential use as a building block in various chemical processes. The discovery and synthesis of 3-phenyltoluene date back to the early development of aromatic hydrocarbons. It can be synthesized through several methods, including the Friedel-Crafts alkylation of toluene with phenyl halides in the presence of a Lewis acid catalyst, such as aluminum chloride. This method is favored for its efficiency and relatively straightforward approach, allowing for the selective formation of the 3-phenyltoluene isomer. The physical properties of 3-phenyltoluene, such as its melting point, boiling point, and solubility in organic solvents, make it a valuable compound in both laboratory and industrial settings. It is a colorless liquid at room temperature and has a distinctive aromatic odor. Its solubility in organic solvents, such as benzene and toluene, facilitates its use in various chemical reactions and processes. One of the primary applications of 3-phenyltoluene is as an intermediate in organic synthesis. Its aromatic structure and functional groups make it a versatile precursor in the production of more complex molecules. For instance, it can undergo electrophilic substitution reactions, allowing for the introduction of various substituents onto the aromatic ring. This reactivity is particularly useful in the synthesis of pharmaceuticals, agrochemicals, and specialty chemicals, where specific functional groups need to be selectively introduced onto an aromatic framework. In addition to its role as a synthetic intermediate, 3-phenyltoluene is also utilized in the development of advanced materials. The compound's aromatic nature contributes to the rigidity and thermal stability of polymers when incorporated into polymer backbones. As a result, it has been explored as a monomer or comonomer in the production of high-performance polymers and resins, which are used in coatings, adhesives, and electronic materials. Furthermore, 3-phenyltoluene has found applications in the field of organic electronics. Its conjugated aromatic system allows it to participate in the formation of organic semiconductors, which are essential components in organic light-emitting diodes (OLEDs), organic photovoltaics (OPVs), and organic field-effect transistors (OFETs). The compound's ability to modify the electronic properties of these materials has made it a valuable asset in the design and optimization of organic electronic devices. In summary, the discovery and application of 3-phenyltoluene have had a significant impact on various fields of chemistry and material science. Its role as a versatile intermediate in organic synthesis, as well as its contribution to the development of advanced materials and organic electronics, underscores its importance in both research and industrial applications. As ongoing research continues to explore new uses and applications for this compound, 3-phenyltoluene is likely to remain a key player in the synthesis of complex molecules and the development of innovative materials. References 1981. Mass fragmentographic determination of polymethylbiphenyl in foods contaminated with petroleum products. Bulletin of Environmental Contamination and Toxicology. DOI: 6894880 2014. An Efficient Pd/Al(OH)3 Nanoparticle Catalyst for Suzuki Coupling Reactions of Aryl Halides. Synthesis. DOI: 10.1055/s-0033-1341084 2023. Synthesis and structural characterization of unsymmetrical Schiff base 2-(1-(2-aminophenylimino)ethyl)phenol (LH3) and its Pd(II) complex [Pd3(LH)3]: catalysis of Suzuki-Miyaura cross-coupling reaction. Journal of Chemical Sciences, 135(2). DOI: 10.1007/s12039-023-02142-9 |
| Market Analysis Reports |