Online Database of Chemicals from Around the World
| Shanghai Scientia Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.scientiapharm.net | |||
 | +86 (21) 5430-1573 6172-1573 400-036-1573 | |||
 | +86 (21) 5186-1981 | |||
 | sales@scientiapharm.com scientiapharm@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Shandong Heyi Gas Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.heyigas.com | |||
 | +86 (546) 236-9006 | |||
 | +86 (546) 236-9003 | |||
 | sales@heyigas.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Shandong Holon Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.holonchem.com | |||
 | +86 (533) 211-1311 | |||
 | +86 (533) 211-1311 | |||
 | admin@holonchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Wuhan Air Gas Electronic Materials Enterprise Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.whagem.com | |||
 | +86 18164072249 | |||
 | s5@whagem.com | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Cambridge Isotope Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.isotope.com | |||
 | +1 (978) 749-8000 | |||
 | +1 (978) 749-2768 | |||
 | cilsales@isotope.com | |||
| Chemical manufacturer | ||||
| Advance Research Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.fluoridearc.com | |||
 | +1 (918) 266-6789 | |||
 | +1 (918) 266-6796 | |||
 | sales@fluoridearc.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal halides and halides >> Metal fluorides and salts |
|---|---|
| Name | Boron trifluoride |
| Synonyms | trifluoroborane |
| Molecular Structure | 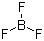 |
| Molecular Formula | BF3 |
| Molecular Weight | 67.80 |
| CAS Registry Number | 7637-07-2 |
| EC Number | 231-569-5 |
| SMILES | B(F)(F)F |
| Density | 1.0$+/-$0.1 g/cm3 Calc.*, 3.076 g/mL (Expl.) |
|---|---|
| Melting point | -127 $degree$C (Expl.) |
| Boiling point | -100.0 $degree$C 760 mmHg (Calc.)*, -100 $degree$C (Expl.) |
| Flash point | 4 $degree$C (Expl.) |
| Solubility | 105-107 g/100g (Expl.) |
| Index of refraction | 1.138 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS04;GHS05;GHS06 Danger Details GHS04;GHS05;GHS06 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H330 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P264-P271-P280-P284-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P320-P321-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1008 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Boron trifluoride is a colorless, toxic, and corrosive gas with the chemical formula BF3. It consists of a boron atom covalently bonded to three fluorine atoms in a trigonal planar geometry, giving the molecule a strong electron-deficient character. Boron trifluoride was first prepared in the early 19th century by the reaction of boron compounds with hydrofluoric acid and has since been widely used as a Lewis acid catalyst in chemical synthesis and industrial processes. The molecular structure of BF3 features sp2 hybridization of the central boron atom, resulting in a planar triangular configuration with bond angles of approximately 120°. The electron deficiency at boron allows the molecule to readily accept electron pairs, making it a strong Lewis acid. Its volatility and reactivity require careful handling, as it reacts with water to form boric acid and hydrogen fluoride, which are highly corrosive. Boron trifluoride is extensively used as a catalyst in organic chemistry, particularly in reactions requiring electron pair acceptance. It catalyzes Friedel–Crafts alkylation and acylation reactions, polymerization of olefins, and isomerization of hydrocarbons. In these processes, BF3 forms adducts with nucleophiles, facilitating the generation of carbocations or activating reactants for further chemical transformations. In addition to its catalytic role, BF3 is used in the production of organoboron compounds, which serve as intermediates in pharmaceuticals, agrochemicals, and specialty materials. Its ability to form stable complexes with diols, amines, and ethers allows for controlled synthesis of boron-containing molecules with applications in chemical research and industrial manufacturing. Boron trifluoride is also employed in analytical chemistry as a complexing agent and in the preparation of boron-doped materials. In the semiconductor industry, it is used to introduce boron into silicon wafers, enabling controlled p-type doping for electronic devices. Its reactivity with moisture and nucleophiles requires handling under dry, inert conditions, often using specialized gas delivery systems or complexed forms, such as BF3·etherate, which provides safer and easier handling. Overall, boron trifluoride is a highly reactive, electron-deficient gas with significant utility as a Lewis acid catalyst, reagent for organoboron synthesis, and dopant in semiconductor fabrication. Its trigonal planar structure and strong affinity for electron pairs make it an essential compound in organic, inorganic, and materials chemistry. References 2025. Mechanism of dry eye syndrome improvement by Tetraselmis chuii: Anti-inflammatory effects and ocular mucous membrane restoration. Journal of Applied Phycology. DOI: 10.1007/s10811-025-03598-7 2025. Silica-loaded perchloric acid as a catalyst for terpene phenol resins. Iranian Polymer Journal. DOI: 10.1007/s13726-025-01526-7 2025. Spontaneous granulation of tricaprin and trilaurin medium-chain triacylglycerols with added medium- and long-chain species. npj Science of Food. DOI: 10.1038/s41538-025-00511-x |
| Market Analysis Reports |