Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Henan Haohai Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.haohaichem.com | |||
 | +86 (371) 6391-6105 | |||
 | +86 (371) 6393-2448 | |||
 | 3h@haohaichem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| LONGRE Pharmaceuticals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.longrepharm.com | |||
 | +86 (512) 6959-2836 +86 13306132836 | |||
 | +86 (512) 6959-2835 | |||
 | sales@longrepharm.com yuguojun1013@163.com | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Changzhou Comwin Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.comwin-china.com | |||
 | +86 (519) 8661-9955 | |||
 | +86 (519) 8661-3190 | |||
 | info@comwin-china.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical pesticide >> Herbicide >> Herbicide intermediate |
|---|---|
| Name | 2,4-Dichlorobenzoyl chloride |
| Synonyms | DCOC; 2,4-Dichlorobenzene-1-carbonyl chloride |
| Molecular Structure | 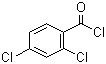 |
| Molecular Formula | C7H3Cl3O |
| Molecular Weight | 209.46 |
| CAS Registry Number | 89-75-8 |
| EC Number | 201-936-4 |
| SMILES | C1=CC(=C(C=C1Cl)Cl)C(=O)Cl |
| Density | 1.49 |
|---|---|
| Melting point | 21 °C |
| Boiling point | 150 °C (34 torr) |
| Refractive index | 1.5917-1.5937 |
| Flash point | 137 °C |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P271-P280-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P319-P321-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2,4-Dichlorobenzoyl chloride is an important chemical compound used in organic synthesis, known for its role as a reactive intermediate in various industrial and pharmaceutical applications. It is an acyl chloride derived from benzoic acid, featuring chlorine atoms at the 2 and 4 positions of the benzene ring. Its molecular formula is C7H3Cl2OCl, and it is a colorless to pale yellow liquid with a pungent odor, typical of acyl chlorides. The compound is highly reactive, particularly in forming amides and esters, making it a key reagent in multiple chemical processes. The discovery of 2,4-Dichlorobenzoyl chloride is connected to research on chlorinated aromatic compounds, which were explored for their potential use in the synthesis of dyes, pharmaceuticals, and agrochemicals. This compound was developed as a specialized reagent due to the reactivity of its acyl chloride group, which allows it to readily participate in nucleophilic acyl substitution reactions. It has become an integral part of the synthetic toolbox for chemists working in both industrial and research settings. One of the primary applications of 2,4-Dichlorobenzoyl chloride is in the agrochemical industry, where it serves as an intermediate in the production of herbicides, fungicides, and insecticides. Its reactivity enables the formation of bioactive compounds that protect crops from pests and diseases. Specifically, it is used to synthesize herbicides that target broadleaf weeds by inhibiting plant growth pathways. The compound’s chlorinated structure enhances the biological activity of the resulting products, contributing to their effectiveness in agricultural applications. In the pharmaceutical field, 2,4-Dichlorobenzoyl chloride is employed in the synthesis of various therapeutic agents. It is particularly useful in the production of anti-inflammatory and antimicrobial drugs, where the acyl chloride group facilitates the introduction of acyl functionalities into core molecular structures. This enhances the pharmacological properties of the compounds, improving their potency and bioavailability. Chlorinated benzoyl derivatives are often sought after for their improved stability and efficacy in drug formulations. Additionally, 2,4-Dichlorobenzoyl chloride finds application in polymer chemistry, where it is used as a cross-linking agent. The reactive acyl chloride group enables the formation of durable bonds in polymer chains, improving the mechanical strength and heat resistance of the materials. This makes the compound valuable in the production of high-performance plastics and coatings that require enhanced durability and stability under harsh conditions. Safety and handling are crucial considerations when working with 2,4-Dichlorobenzoyl chloride, as it is highly corrosive and can release harmful fumes when exposed to moisture. Proper protective equipment and ventilation are necessary during its use, and the compound must be stored in airtight containers to prevent degradation. Environmental concerns related to the use of chlorinated compounds have led to strict regulations governing its disposal and emissions. In summary, 2,4-Dichlorobenzoyl chloride is a versatile and reactive compound with significant applications in agrochemicals, pharmaceuticals, and polymer chemistry. Its ability to participate in acylation reactions makes it a key intermediate in the production of a wide range of bioactive and industrial compounds. However, careful handling and adherence to safety protocols are essential due to its corrosive nature and potential environmental impact. References 2015. Synthesis and blood glucose and lipid-lowering effects of benzothiazole-substituted benzenesulfonylurea derivatives. Monatshefte für Chemie - Chemical Monthly, 146(6). DOI: 10.1007/s00706-015-1471-2 2011. Selective Mg Insertion into Substituted Mono- and Dichloro Arenes in the Presence of LiCl: A New Preparation of Boscalid. Synlett, 2011(16). DOI: 10.1055/s-0030-1261168 2008. 2,4-Dichloro-N-cyclohexylbenzamide. Acta crystallographica. Section E, Structure reports online, 64(4). DOI: 10.1107/s1600536808008131 |
| Market Analysis Reports |