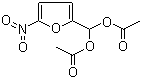
5-Nitro-2-furaldehyde diacetate is an organic compound that consists of a furan ring with a nitro group and an aldehyde group at the 5 and 2 positions, respectively. The compound is further acetylated with two acetate groups, one at the aldehyde position and one at the hydroxyl group attached to the furan ring. This structure combines functional groups that are reactive and useful in various synthetic applications.
The discovery of 5-nitro-2-furaldehyde diacetate is tied to the study of functionalized furfural derivatives, which are of interest due to their reactivity and versatility in organic synthesis. Furfural and its derivatives have long been used as intermediates in the synthesis of a wide range of chemicals, particularly in the fields of pharmaceuticals and agrochemicals. The nitro and acetyl functional groups attached to the furan ring in 5-nitro-2-furaldehyde diacetate modify its reactivity, offering new possibilities for chemical transformations.
5-Nitro-2-furaldehyde diacetate is primarily utilized in organic synthesis as a reactive intermediate. The nitro group provides an electron-withdrawing effect that enhances the electrophilic nature of the aldehyde, making it more susceptible to nucleophilic attack. This characteristic is valuable in a variety of reactions, including those leading to the formation of carbon-carbon bonds. Its reactivity allows for the formation of various derivatives through condensation reactions, where it can react with nucleophiles such as amines, alcohols, or thiols. This makes the compound useful in the synthesis of more complex organic molecules.
Additionally, the compound’s application extends to the field of material science. The acetyl groups can participate in esterification reactions, and the furan ring’s ability to engage in polymerization reactions makes 5-nitro-2-furaldehyde diacetate a useful precursor for the creation of specialized polymers. Such polymers may have applications in coatings, adhesives, and other materials where furan-based chemistry is advantageous due to its stability and functional diversity.
Another application of 5-nitro-2-furaldehyde diacetate is in the preparation of biologically active molecules. As a building block in medicinal chemistry, it can serve as a precursor for the synthesis of potential pharmaceuticals. For example, the compound can be incorporated into molecules designed for antimicrobial or anticancer activity, as the nitro group and furan ring structure contribute to the molecular properties that interact with biological systems.
In conclusion, 5-nitro-2-furaldehyde diacetate is a compound with several applications in organic synthesis, material science, and medicinal chemistry. Its functional groups offer significant reactivity, allowing it to serve as an intermediate for the formation of more complex molecules with potential use in various industrial and pharmaceutical applications. The well-established reactivity of furan derivatives in chemical reactions underpins the value of 5-nitro-2-furaldehyde diacetate in these fields.
References
2001. Synthesis of 2-Cyano-5-nitrofuran. Chemistry of Heterocyclic Compounds, 37(9).
DOI: 10.1023/a:1013204422043
1993. Biological activity of furan derivatives (review). Chemistry of Heterocyclic Compounds, 29(3).
DOI: 10.1007/bf00531499
1990. The application of levulinic acid and 5-nitro-2-furylmethylene diacetate in the total synthesis of some novel biologically active (5-nitro-2-furyl)azomethines. Monatshefte für Chemie / Chemical Monthly, 121(11).
DOI: 10.1007/bf00808957
|






































 GHS07;GHS08 Warning Details
GHS07;GHS08 Warning Details