Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8816-2785 +86 13606544505 | |||
 |
lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink massive supplier since 2021 | ||||
| Changzhou Huaren Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (519) 8812-2965 | |||
 |
huarenchem@vip.163.com sales@huarenchem.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink standard supplier since 2006 | ||||
| Hangzhou Nanbo Biochemical Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8873-9652 +86 15355716006 | |||
 |
sales@nanbobiochem.com emiliy_88@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| Simagchem Corporation | China | Inquire | ||
|---|---|---|---|---|
 |
+86 13806087780 | |||
 |
sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink standard supplier since 2008 | ||||
| Shanghai Growingchem Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (21) 5080-0795 | |||
 |
sales@growingchem.com syzhang1983@163.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink standard supplier since 2009 | ||||
| Shanghai Yurlic Chemical S&T Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (21) 37285141 +86 15000957566 | |||
 |
yurlichem@hotmail.com sales@yurlichem.cn | |||
 |
QQ chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink standard supplier since 2009 | ||||
| Ring Specialty Chemicals Inc. | Canada | Inquire | ||
|---|---|---|---|---|
 |
+1 (416) 493-6870 | |||
 |
info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (609) 683-1117 | |||
 |
Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink standard supplier since 2010 | ||||
| Classification | Organic raw materials >> Alcohols, phenols, phenolic compounds and derivatives |
|---|---|
| Name | (S)-(+)-2,2-Dimethyl-1,3-dioxolane-4-methanol |
| Synonyms | (+)-2,3-O-Isopropylidene-sn-glycerol; (S)-(+)-Solketal |
| Molecular Structure | 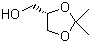 |
| Molecular Formula | C6H12O3 |
| Molecular Weight | 132.16 |
| CAS Registry Number | 22323-82-6 |
| EC Number | 244-910-8 |
| SMILES | CC1(OC[C@@H](O1)CO)C |
| Density | 1.0±0.1 g/cm3 Calc.*, 1.066 g/mL (Expl.) |
|---|---|
| Boiling point | 188.5 ºC 760 mmHg (Calc.)*, 215.9 - 217.3 ºC (Expl.) |
| Flash point | 80.0 ºC (Calc.)*, 79 ºC (Expl.) |
| Solubility | water, miscible (Expl.) |
| Index of refraction | 1.424 (Calc.)*, 1.434 (Expl.) |
| Alpha | 11 º (c=5, CH3OH) (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |

 GHS07 WarningGHS02; Details
GHS07 WarningGHS02; Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hazard Statements | H227-H315-H319-H335 Details | ||||||||||||||||||||||||||||
| Precautionary Statements | P210-P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P370+P378-P403-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
(S)-(+)-2,2-Dimethyl-1,3-dioxolane-4-methanol is a chiral acetal derivative belonging to the class of 1,3-dioxolane compounds, which are widely used in organic synthesis as protecting groups for diols and carbonyl compounds. This substance contains a five-membered cyclic acetal ring formed from a diol and acetone, with a hydroxymethyl substituent at the 4-position and two methyl groups at the 2-position. Because of its defined stereochemistry and stability under neutral and basic conditions, it is frequently used as an intermediate in carbohydrate chemistry, pharmaceutical synthesis, and stereoselective organic reactions. The structure of (S)-(+)-2,2-dimethyl-1,3-dioxolane-4-methanol consists of a 1,3-dioxolane ring containing two oxygen atoms separated by a methylene bridge, with the 2-position bearing two methyl groups derived from acetone. The 4-position carries a hydroxymethyl group, and this carbon is chiral, giving rise to the (S) configuration indicated in the name. The (+) designation refers to the positive optical rotation observed for this enantiomer. The cyclic acetal framework provides protection for adjacent hydroxyl groups and can be removed under acidic conditions, making the compound useful in multistep synthesis. The preparation of this compound is commonly achieved by acetalization of glyceraldehyde or related diols with acetone in the presence of an acid catalyst, followed by reduction or functional group modification to obtain the hydroxymethyl substituent in the desired configuration. Control of stereochemistry is important during synthesis, since the chiral center determines the optical activity and suitability of the compound for use in enantioselective reactions. The product is typically obtained as a stable liquid or low-melting solid that can be stored under normal laboratory conditions. In carbohydrate chemistry, 2,2-dimethyl-1,3-dioxolane derivatives are widely used as protecting groups for vicinal diols. The acetonide group formed by reaction with acetone protects two hydroxyl groups simultaneously while maintaining a rigid cyclic structure. This protection strategy allows selective reactions at other positions in sugars or polyols, which is essential for the synthesis of nucleosides, glycosides, and other biologically important molecules. The hydroxymethyl substituent in this compound provides an additional functional handle that can be oxidized, reduced, or converted into other groups without disturbing the acetal ring under appropriate conditions. Chiral dioxolane derivatives such as (S)-(+)-2,2-dimethyl-1,3-dioxolane-4-methanol are also used as intermediates in the preparation of pharmaceuticals and fine chemicals. The defined stereochemistry allows them to serve as starting materials for the synthesis of optically active compounds, including amino alcohols, heterocycles, and enzyme inhibitors. Because many biological systems are sensitive to stereochemistry, the availability of enantiomerically pure intermediates is essential for producing compounds with the desired activity and minimal side effects. In addition to their use in synthesis, dioxolane acetals are valued for their chemical stability and predictable reactivity. They resist hydrolysis under neutral or basic conditions but can be cleaved in the presence of acid to regenerate the original diol or carbonyl compound. This property makes them especially useful in stepwise synthetic sequences where temporary protection of functional groups is required. The combination of a stable acetonide ring, a reactive hydroxymethyl group, and defined chirality makes (S)-(+)-2,2-dimethyl-1,3-dioxolane-4-methanol an important intermediate in modern organic chemistry. References 2022. 1-(6-Methylsalicyloyl)glycerol from stony coral-derived Micromonospora sp. The Journal of Antibiotics. DOI: 10.1038/s41429-022-00578-8 2020. Stereoselective 1,4-Addition of Primary Alcohols to ?-Alkoxy-a,�-unsaturated Esters. Synthesis. DOI: 10.1055/s-0040-1707274 2015. Intramolecular Enantiomerism in S(+)2,2-dimethyl -1,3-dioxolane-4-methanol: The Interpretation of Raman Optical Activity Intensity. Chirality. DOI: 10.1002/chir.22507 |
| Market Analysis Reports |
| List of Reports Available for (S)-(+)-2,2-Dimethyl-1,3-dioxolane-4-methanol |