Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8816-2785 +86 13606544505 | |||
 |
lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink massive supplier since 2021 | ||||
| Changzhou Harvechem Ltd | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (519) 8878-4888 8872-1112 | |||
 |
ppchem@vip.163.com harvechem@vip.163.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| Ningbo Tianyuan Chemistry Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (574) 8821-6239 8821-6587 8303-6872 | |||
 |
sale@tianyuan818.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| Nanjing Pharmatechs Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (25) 5839-4007 | |||
 |
sales@nanjingpharma.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| Simagchem Corporation | China | Inquire | ||
|---|---|---|---|---|
 |
+86 13806087780 | |||
 |
sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink standard supplier since 2008 | ||||
| Shangyu Zhebang Pharmaceutical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (575) 8273-2308 | |||
 |
sales@zbchem.com sales@zbpharm.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2009 | ||||
| Minakem S.A.S. | France | Inquire | ||
|---|---|---|---|---|
 |
+33 (3) 2064-6830 | |||
 |
contact@minakem.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2009 | ||||
| Boron Molecular Inc. | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (919) 981-4441 | |||
 |
sales@boronmolecular-usa.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink standard supplier since 2009 | ||||
| Classification | Chemical reagent >> Organic reagent >> Boric acid |
|---|---|
| Name | Cyclopropylboronic acid |
| Molecular Structure | 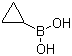 |
| Molecular Formula | C3H7BO2 |
| Molecular Weight | 85.90 |
| CAS Registry Number | 411235-57-9 |
| EC Number | 609-890-0 |
| SMILES | B(C1CC1)(O)O |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 90 - 95 ºC (Expl.) |
| Boiling point | 205.1±23.0 ºC 760 mmHg (Calc.)* |
| Flash point | 77.9±22.6 ºC (Calc.)* |
| Index of refraction | 1.443 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |



 GHS02;GHS05;GHS07;GHS08 Danger Details
GHS02;GHS05;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hazard Statements | H242-H302-H315-H318-H319-H335-H360 Details | ||||||||||||||||||||||||||||||||||||||||
| Precautionary Statements | P203-P210-P234-P235-P240-P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P317-P318-P319-P321-P330-P332+P317-P337+P317-P362+P364-P370+P378-P403-P403+P233-P405-P410-P411-P420-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Cyclopropylboronic acid is an organoboron compound in which a cyclopropyl group is directly attached to a boronic acid functional group, B(OH)2. Boronic acids are widely used in organic synthesis because they participate in carbon–carbon bond forming reactions, especially palladium-catalyzed cross-coupling processes. The presence of the strained cyclopropyl ring gives this compound additional synthetic value, since cyclopropyl groups are frequently introduced into pharmaceutical molecules, agrochemicals, and functional materials to modify biological activity, stability, or molecular conformation. The structure of cyclopropylboronic acid contains a three-membered cyclopropane ring bonded to a boron atom that is also attached to two hydroxyl groups. The boron atom is electron-deficient and can form reversible covalent bonds with nucleophiles such as alcohols, amines, or diols. Because of this property, boronic acids are useful intermediates in many types of reactions, including ester formation, oxidation, and cross-coupling. The cyclopropyl group is characterized by high ring strain and strong σ bonding, which can influence the reactivity and stereochemical outcome of reactions in which the compound participates. The preparation of cyclopropylboronic acid is commonly achieved through metal–halogen exchange or lithiation of cyclopropyl derivatives followed by reaction with trialkyl borates and subsequent hydrolysis. Another widely used method involves palladium-catalyzed borylation of cyclopropyl halides using diboron reagents under controlled conditions. These procedures allow the boronic acid to be obtained in good yield while preserving the integrity of the strained cyclopropyl ring. The product is usually isolated as a crystalline solid or as a stable solution that can be handled under normal laboratory conditions. One of the most important applications of cyclopropylboronic acid is in Suzuki–Miyaura cross-coupling reactions. In this reaction, the boronic acid reacts with an aryl or vinyl halide in the presence of a palladium catalyst and a base to form a new carbon–carbon bond. This method is widely used in the synthesis of pharmaceuticals and fine chemicals because it proceeds under relatively mild conditions and tolerates many functional groups. The cyclopropyl group introduced by this reaction can significantly influence the biological properties of the final molecule by changing its shape, lipophilicity, and metabolic stability. Cyclopropyl-substituted compounds are often used in medicinal chemistry because the small, rigid ring can mimic double bonds or bulky substituents while resisting metabolic oxidation. For this reason, cyclopropylboronic acid is frequently employed as a building block for the preparation of kinase inhibitors, antiviral agents, and other biologically active molecules. The boronic acid functionality allows the cyclopropyl group to be introduced at a late stage in synthesis, which is advantageous when complex molecules are being prepared. In addition to pharmaceutical synthesis, cyclopropylboronic acid is used in the preparation of advanced materials and ligands for catalysis. Boronic acids can form reversible complexes with diols and other Lewis bases, which is useful in sensing, polymer chemistry, and supramolecular design. The presence of the cyclopropyl group can alter steric and electronic properties, allowing chemists to fine-tune the behavior of the resulting compounds. Because of its combination of boron reactivity and strained ring structure, cyclopropylboronic acid remains an important intermediate in modern organic chemistry. References 2025. A Greener and Scalable Approach for the Synthesis of MRT67307 and Its AntiInflammatory Activity. Russian Journal of General Chemistry. DOI: 10.1134/s1070363224613474 2023. Metal-catalysed C�C bond formation at cyclopropanes. Nature reviews. Chemistry. DOI: 10.1038/s41570-023-00499-6 2020. Synthetic Approaches to Contemporary Drugs that Contain the Cyclopropyl Moiety. Synthesis. DOI: 10.1055/s-0039-1690058 |
| Market Analysis Reports |
| List of Reports Available for Cyclopropylboronic acid |